Authors:
Huihui Wang, Wu Zeng, Kenneth Munge Kabubei, Jennifer Rasanathan, Jacob Kazungu, Sandile Ginindza, Sifiso Mtshali, Luis E Salinas, Amanda McClelland, Marine Buissonniere, Christopher T Lee, Jane Chuma, Jeremy Veillard, Thulani Matsebula, Mickey Chopra
Publication date: 2023
© 2023 International Bank for Reconstruction and Development / The World Bank
1818 H Street NW
Washington DC 20433
Telephone: 202-473-1000
Internet: www.worldbank.org
This work is a product of the staff of The World Bank with external contributions. The findings, interpretations, and conclusions expressed in this work do not necessarily reflect the views of The World Bank, its Board of Executive Directors, or the governments they represent.
The World Bank does not guarantee the accuracy, completeness, or currency of the data included in this work and does not assume responsibility for any errors, omissions, or discrepancies in the information, or liability with respect to the use of or failure to use the information, methods, processes, or conclusions set forth. The boundaries, colors, denominations, and other information shown on any map in this work do not imply any judgment on the part of The World Bank concerning the legal status of any territory or the endorsement or acceptance of such boundaries.
Nothing herein shall constitute or be construed or considered to be a limitation upon or waiver of the privileges and immunities of The World Bank, all of which are specifically reserved.
Rights and Permissions
The material in this work is subject to copyright. Because The World Bank encourages dissemination of its knowledge, this work may be reproduced, in whole or in part, for noncommercial purposes as long as full attribution to this work is given.
Any queries on rights and licenses, including subsidiary rights, should be addressed to World Bank Publications, The World Bank Group, 1818 H Street NW, Washington, DC 20433, USA; fax: 202-522-2625; e-mail: pubrights@worldbank.org.
Introduction
Health care workers (HCWs) face disproportionate risk of exposure and becoming ill in any infectious disease outbreak. SARS-CoV-2 has proven no exception: from Wuhan to Manaus, London to Tehran, and Delhi to Johannesburg, HCWs working in clinics and hospitals have been at heightened risk of developing COVID-19 disease, especially at the beginning of the pandemic when little was known about the pathogen. The estimated incidence of SARS-CoV-2 infection amongst HCWs in the first year of the pandemic varied widely across settings, from less than 1% to nearly 50% positivity on PCR testing, with more recent evidence suggesting an average incidence of 12.5% amongst HCWs in China, the US and Europe.
High rates of HCW infection and death exacerbate the health impacts of epidemics and pandemics through many channels. First, infected HCWs can be important vectors of onward disease transmission to patients and colleagues, amplifying or seeding broader community outbreaks. This was demonstrated with SARS: infections acquired in health care settings accounted for the majority of SARS cases in Singapore, Toronto, and Taiwan. Health care–acquired SARS-CoV-2 infections have similarly proven to be prevalent, more so before the development of COVID-19 vaccines. HCWs with SARS-CoV-2 infection are also important sources of viral transmission to other HCWs and to household members.
Second, HCW absenteeism due to COVID-19 illness, isolation or death, and fear of being infected disrupts essential health service delivery. It is now widely understood that health systems overwhelmed by surging COVID-19 cases are not only compromised in their ability to care for those with COVID-19 disease, but also for patients with other life-threatening emergencies or chronic health care needs. Essential health care services including maternity care, immunization, chronic disease management, emergency services, and surgery were severely disrupted in the first year of the pandemic, especially in low- and middle-income countries (LMICs), where human resources for health are already in chronically short supply. For example, an estimated 8.74 million patient visits across nine health services in Mexico were not done in the first nine months of the pandemic, and more than 300,000 doses of an essential pediatric vaccine were missed across eight countries in sub-Saharan Africa in the first five months of the pandemic. Detrimental impacts of COVID-19 have been documented on maternal and child health care globally: fewer immunizations were given, fewer women received the full scope of antenatal care, and fewer babies were delivered in health care facilities. Studies reveal stark differences in maternal and child health service provision due to the COVID-19 pandemic.
As a result, child and maternal death rates were expected to increase and already have in some countries. Maternal and child health care delivery is directly compromised by HCW illness and deaths; fear of acquiring infections in healthcare settings, lack of trust in public health care services, and even movement restrictions often stem from HCW infections and further deter patients from seeking care.
This study aims to estimate the economic costs of SARS-CoV-2 infections in HCWs during the first year of the pandemic from the societal perspective in four low- or middle-income countries. We propose a framework to translate SARS-CoV-2 infection amongst HCWs into economic costs along three pathways, provide the estimated burden of HCW infections, and offer recommendations to mitigate against future economic losses due to HCW infections. The economic burden due to SARS-CoV-2 infection among HCWs makes a compelling investment case for pandemic preparedness, particularly the protection of HCWs and resilient health systems going forward.
It is critical to understand the burden of COVID-19 attributable to HCW infections. Though it is well-known and widely understood that COVID-19 is not just a health crisis, but an economic crisis,40 the extent to which HCW infections and deaths due to COVID-19 pose a society-wide economic burden is less well understood. The burden of illness or death of HCWs includes costs of medical care, diminished personal earnings, and lost economic productivity over time.41 These costs are amplified by greater infectious spread, including in health care settings, within households of HCWs and to the wider community. Further, the economic costs associated with disruptions in health care service delivery when HCWs are sick are borne by the entire society.41,42
Studies have sought to quantify the overall economic burden of the COVID-19 pandemic43, including the cost of lost productivity due to premature deaths from
COVID-1944, without specifically examining the economic burden attributed to COVID infections and deaths in HCWs. One study in Iran estimated the economic costs of SARS-CoV-2 infections in HCWs, with a focus on productivity losses due to HCW absenteeism and medical costs of SARS-CoV-2 infection in HCWs. Similar to studies on the economic burden of illness, examining the economic burden (cost) of SARS-CoV-2 infections among HCWs would help raise the awareness of COVID-19 infections in this specific group of professionals during the pandemic and prioritize interventions to address it. To our knowledge, there is no empirical evidence of the total society-wide economic burden of SARS-CoV-2 infections in HCWs, especially in LMICs, capturing the direct and indirect cost of HCW infections, their contributions to wider community transmissions and the economic toll of disrupted health services. Understanding the greater scale of economic costs may move national decision-makers and the global health community beyond panic-neglect cycles in pandemic response financing toward (re-)building a resilient health workforce and more sustainable, enduring pandemic preparedness, which includes adequate protection of HCWs. This study aims to estimate the economic costs of SARS-CoV-2 infections in HCWs during the first year of the pandemic from the societal perspective in four low- or middle-income countries. We propose a framework to translate SARS-CoV-2 infection amongst HCWs into economic costs along three pathways, provide the estimated burden of HCW infections, and offer recommendations to mitigate against future economic losses due to HCW infections. The economic burden due to SARS-CoV-2 infection among HCWs makes a compelling investment case for pandemic preparedness, particularly the protection of HCWs and resilient health systems going forward.
Chapter 1. Data and Methodology
I. Overall approach
We developed a framework that lays out the major pathways through which SARS-CoV-2 infection in HCWs leads to morbidity and mortality across the population based on a review of the literature regarding the health and economic impacts of SARS-CoV-2 infection in HCWs. To quantify the economic burden of COVID-19 attributable to HCW infection, we follow the traditional cost-of-illness (COI) approach to translate additional morbidity and mortality into the economic burden on a societal level (Figure 1). The COI approach, which focuses on the economic consequences of diseases, was widely used in studies of the economic burden of diseases, such as diabetes, dengue, and respiratory diseases. Generally, the COI approach concentrates on therapeutic costs and the cost associated with productivity loss due to illness. It does not estimate the cost of interventions to prevent the diseases.
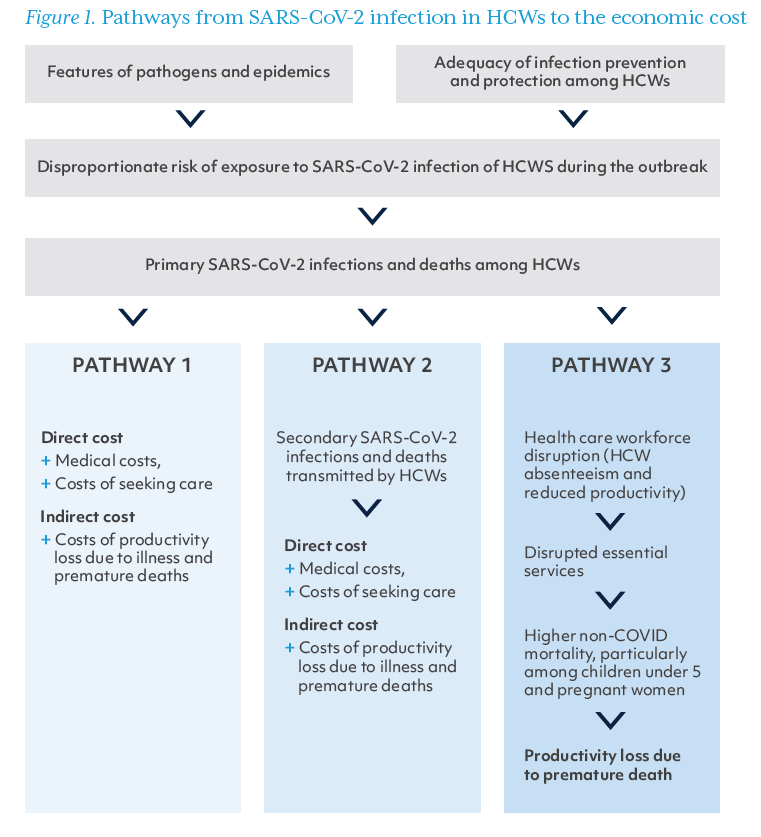
The framework focuses on three pathways through which SARS-CoV-2 infection in HCWs leads to economic costs to society:
1. This first pathway, also the most direct pathway, accounts for the costs associated with primary infections and deaths among HCWs. HCWs in this study are defined as those who work in health institutions in professional health-related positions, including physicians, nurses, lab technicians, and health administrators (excluding cleaners, drivers, and community health workers). Ill HCWs need to seek care, receive treatment, and take time off from work for recovery and isolation. We captured two types of costs for this pathway: (a) direct costs, i.e., costs directly associated with care-seeking due to COVID-19, including both medical costs and non-medical costs (e.g., meals and transportation); and (b) indirect costs, estimated with the human capital approach, i.e., the costs associated with lost productivity while sick and recovering or, in the case of death, for the years of economic productivity lost relative to the life expectancy.
2.This second pathway accounts for the costs of secondary infections and deaths in the general population attributable to transmission from infected HCWs. As described above, HCWs can drive disease transmission. Infections and deaths in this context refer especially to infections and deaths among those who have close contact with HCWs, for example, household members and patients. The direct and indirect costs included under Pathway 2 are estimated in the same way as for Pathway 1.
3. The third pathway encompasses the economic cost of excess deaths due to conditions other than COVID-19 as a result of health care workforce disruptions (e.g., absenteeism and reduced productivity due to stress, fatigue or missing essential team members). These costs largely result from lost human capital and lost years of economic productivity due to premature deaths. The full scope of health consequences due to HCW infection–mediated disruptions in service delivery is not yet known, but there is already compelling evidence of impact on child and maternal mortality due to health workforce shortages. In this pathway, we used the human capital approach to estimate the productivity losses due to additional maternal and child deaths.
In this study, we estimated the economic costs incurred due to HCW infection during the first year of the COVID-19 pandemic through each of these three pathways and in total in Eswatini, Kenya, Colombia, and two provinces of South Africa. In South Africa, we focused on KwaZulu-Natal and Western Cape provinces because national aggregates were not available and these two provinces accounted for 40% of the national COVID-19 burden by February 2021. The study period was between March 1, 2020 and February 28, 2021.
II. Methods
Table 1 summarizes the population groups included as well as the economic costs captured in each pathway using the COI approach. In the following section, we explain how we estimated the number of cases and deaths in each group and present formulas for estimating each type of cost.
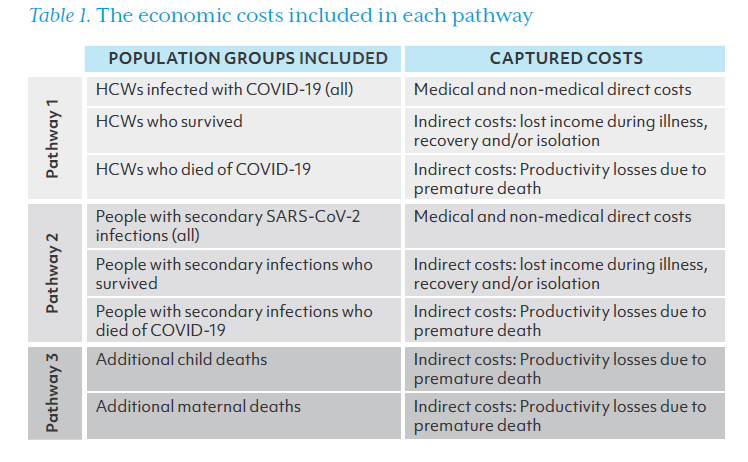
Estimating infections and deaths under each pathway
PATHWAY 1:
The number of SARS-CoV-2 infections in HCWs, as well as the number of HCWs who either survived or died, was collected through primary data collection by the research team from country data sources described in Annex 1 and presented in the results section.
PATHWAY 2:
The number of people with secondary infections was estimated by applying the concept of population attributable risk (PAR), the proportion of SARS-CoV-2 infections in the general population that was attributable to close contacts of HCWs in each study site. In this study, we defined close contacts as HCWs’ household members and patients admitted to hospitals for inpatient care. The household size for each country was obtained from government documents, and we assumed 20% of inpatients as close contacts of HCWs in the main analysis. The number of 20% is based on the contact intensity of inpatients with HCWs compared to that of HCWs’ family members with HCWs. The number of secondary infections was estimated to be the product of total SARS-CoV-2 infections and PAR. We then estimated the number of deaths due to secondary infections to be the product of the number of secondary infections and the case fatality rate in each site. PAR was calculated using the following formula:
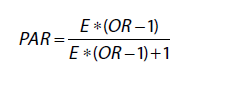
where E is the share of the population considered to be close contacts of HCWs, and OR is the odds ratio of being diagnosed with COVID-19 due to HCW exposure. Annex 2 presents detailed estimations of E and OR.
PATHWAY 3:
To estimate the additional child deaths resulting from SARS-CoV-2 infection and death in HCWs, we first converted the duration of HCW absence from work and the associated reduction in productivity into a decrease in HCW density. For HCWs who were not infected with SARS-CoV-2, we assumed a 10% reduction in their working productivity absent essential team members, using the same assumption as Roberton’s study in the main analysis.

We estimated the resulting change in the under-five mortality rate (U5MR) based on the elasticity of U5MR relative to HCW density,55 and from this calculated additional deaths in children under five years old.
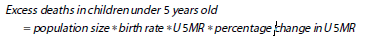
Where percentage change in U5MR = elasticity * percentage reduction in HCW density We applied the same method to estimate excess maternal deaths, using the maternal mortality rate (MMR) and its elasticity relative to HCW density.
ESTIMATING DIRECT COSTS
Direct cost estimation applies to all COVID 19 cases in pathways 1 and 2 (both survived and lethal cases) and includes two elements: medical costs and non-medical cost. The medical costs were estimated to be:

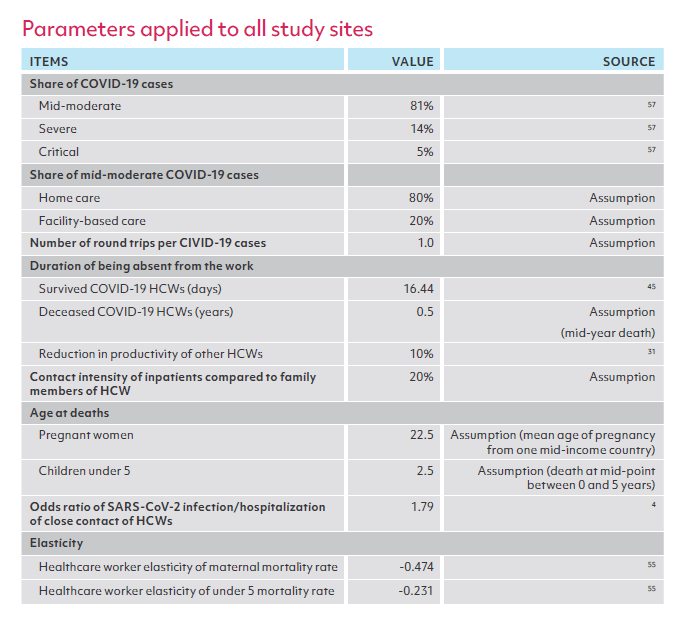
where d is the severity of COVID-19 disease (e.g., mild-moderate, severe, or critical). In this study, we assumed that 81% of non-lethal SARS-CoV-2 infections were mild to moderate, 14% were severe and 5% were critical in severity. Of lethal cases, we assumed that 0.85% resulted from mild-moderate disease, 1.7% derived from severe disease, and the vast majority, 97.45%, resulted from critical cases. The local treatment cost per case at each severity level was obtained from peer-reviewed literature or grey literature. Barasa et al. presented the treatment costs of COVID-19 disease in Kenya by severity level. Where treatment costs were not available for all severity levels, , we derived them using the known corresponding cost ratio from Kenya (Annex 1). For example, it was estimated the ratio of treatment cost of the severe COVID-19 case to that of the mild-moderate hospitalized case was estimated to be 1.69 based on collected data in Kenya. We applied this ratio to estimating the treatment cost of the mild-moderate hospitalized case in Eswatini, as the associated cost was not available, but the cost of the severe case could be obtained in Eswatini.
The non-medical costs of HCW infections include travel and meal costs while seeking or receiving facility-based care.
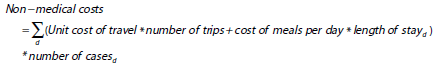
where d is the severity of COVID-19 disease (e.g., mild-moderate, severe, or critical). We assumed one trip was needed per COVID-19 episode regardless of the severity.
ESTIMATING INDIRECT COSTS
For those who were infected with SARS-CoV-2 and survived, the indirect costs of their illness were estimated to be their lost income for the period when they couldn’t work due to COVID-19 disease.

To estimate the indirect costs for those who died of COVID-19, we used the human capital approach to estimate the productivity losses associated with premature death. Gross Domestic Product (GDP) per capita in each country was used as a proxy for annual productivity loss.42 Total productivity loss was estimated as the present value of lost productivity across all anticipated productive years lost. We estimated the number of productive years lost based on life expectancy at the age of death according to the Global Health Observatory.
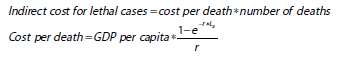
where r is the annual discount rate (3%), e is the exponential constant and La is life expectancy at the age of death.
III. Site selection
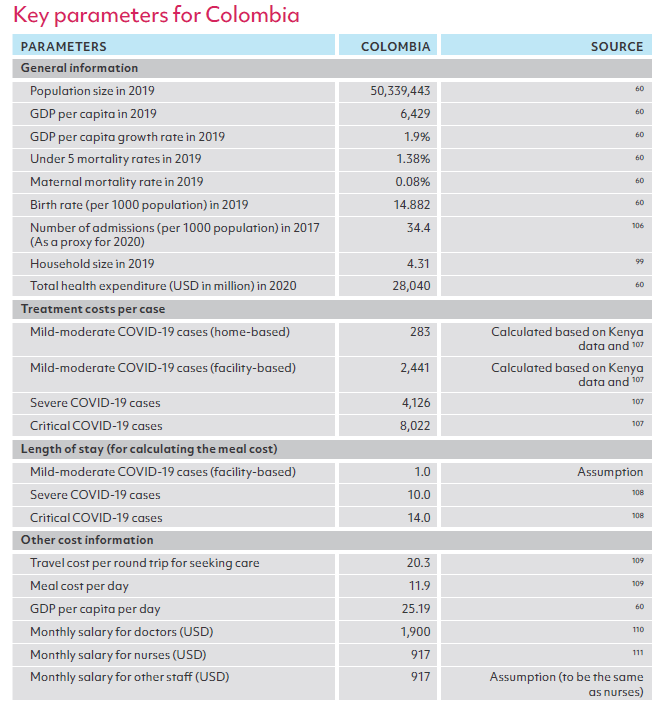
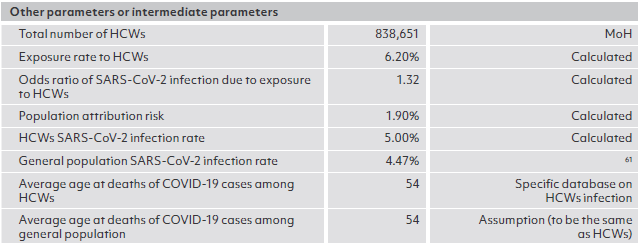
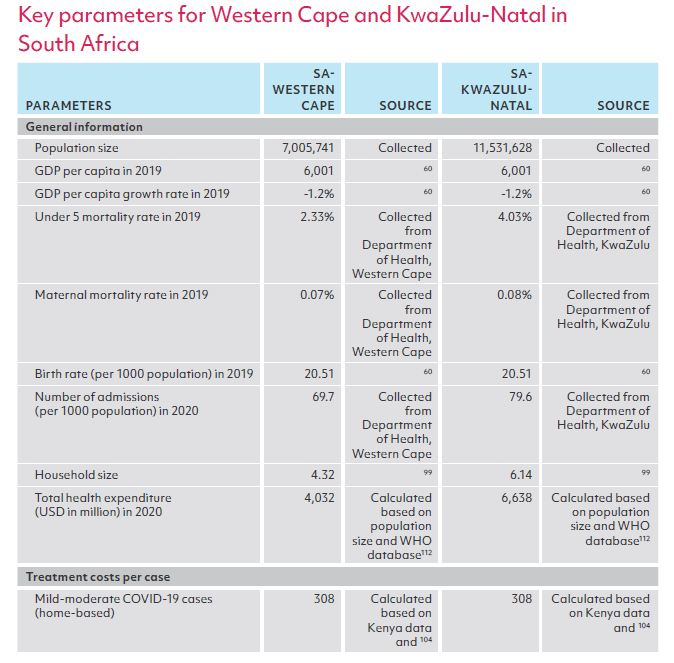
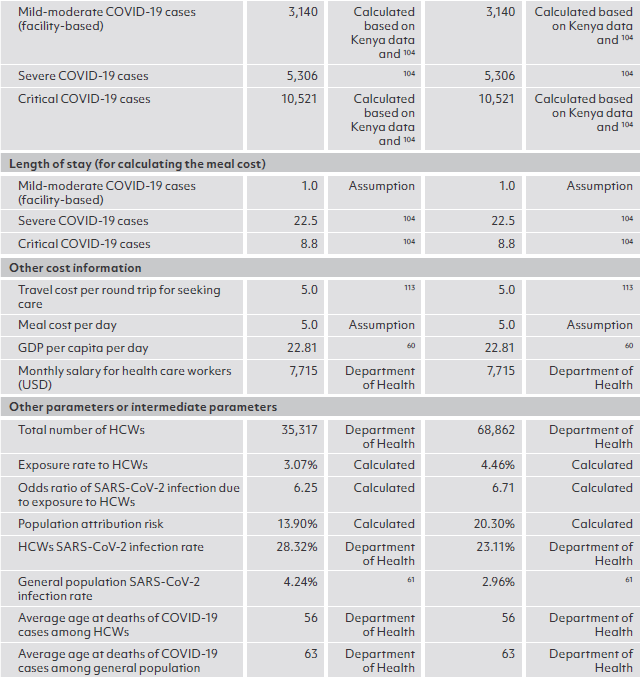
Taking into consideration the feasibility of conducting the study and demonstrating the economic cost of HCW infection in various country settings, we selected five study sites (Kenya, Eswatini, Colombia, and Western Cape province and KwaZulu-Natal province in South Africa). The study sites had diverse profiles with respect to their overall demographics, HCW density, SARS-CoV-2 infection, and death rates in both HCWs and the general population. Table 2 summarizes the patterns of relevant key indicators for each site. For example, Colombia is a populous country with more human resources for health. Although the SARS-CoV-2 infection and mortality rates in the general population were high, HCWs had only a slightly higher infection rate than the general population, with a very low mortality rate. Kenya, another populous country, presented a divergent profile: low HCW density, low infection, and mortality rates in the general population, but HCWs faced a much higher risk of infection. KwaZulu-Natal and Western Cape were characterized by high overall infection rates with an even higher infection rate in HCWs.

IV. Data Sources
To estimate the costs of HCW infections according to the methodology described above, we used a mix of data sources: a) primary data collection in close collaboration with World Bank country offices and national authorities on SARS-CoV-2 infections in HCWs and their outcomes (i.e., survival or death) in the first year of the pandemic, as well as average HCW income by profession to the extent possible; b) World Development Indicators (WDI) on country demographics, macroeconomic figures (e.g., GDP per capita) and health sector characteristics and indicators (e.g., total health expenditure, total government health expenditure, hospital admission rates, under-five mortality rate, maternal mortality rate)60; c) the Johns Hopkins University database on SARS-CoV-2 infections and deaths in the general population;61 d) peer-reviewed journal publications or grey literature for other key parameters such as treatment cost per case, the composition of cases by severity level, length of stay in a medical facility, duration of absence from work; and e) the team’s own assumptions. Annex 1 provides the details on the sources of all data and assumptions used for each study site.
V. Scenario and sensitivity analysis
To understand the sensitivity of the change of parameters to the estimated economic burden, we created three scenarios based on different combinations of values in four parameters which are critical to final cost estimates and more likely subject to a range instead of fixed-point estimates: (1) the share of inpatients considered to be close contacts of HCWs; (2) the extent to which COVID-19 reduced health care workforce productivity; (3) the elasticity of MMR relative to HCW density; and (4) the elasticity of U5MR relative to HCW density.
Table 3 shows the value of these 4 indicators in low, moderate, and high-impact scenarios. We used the moderate impact scenario for the main analysis and presented all findings under this scenario. The high- and low-impact scenarios assumed the independence of the four parameters. The high-impact scenario presented a scenario that would result in a high economic burden of SARS-CoV-2 infection among HCWs by adjusting up the share of inpatients as close contact of HCWs, reduction in HCWs’ productivity, and high elasticities of MMR and U5MR. Conversely, the low-impact scenario adjusted down those parameters. We additionally conducted one-way sensitivity analyses of these four parameters and examined how they affect the economic costs, reported as percentage changes from the main analysis.

Chapter 2. Results
I. Pathway 1: The economic costs of HCW primary infections and deaths due to
COVID-19
I-A. Infections and deaths among HCWs
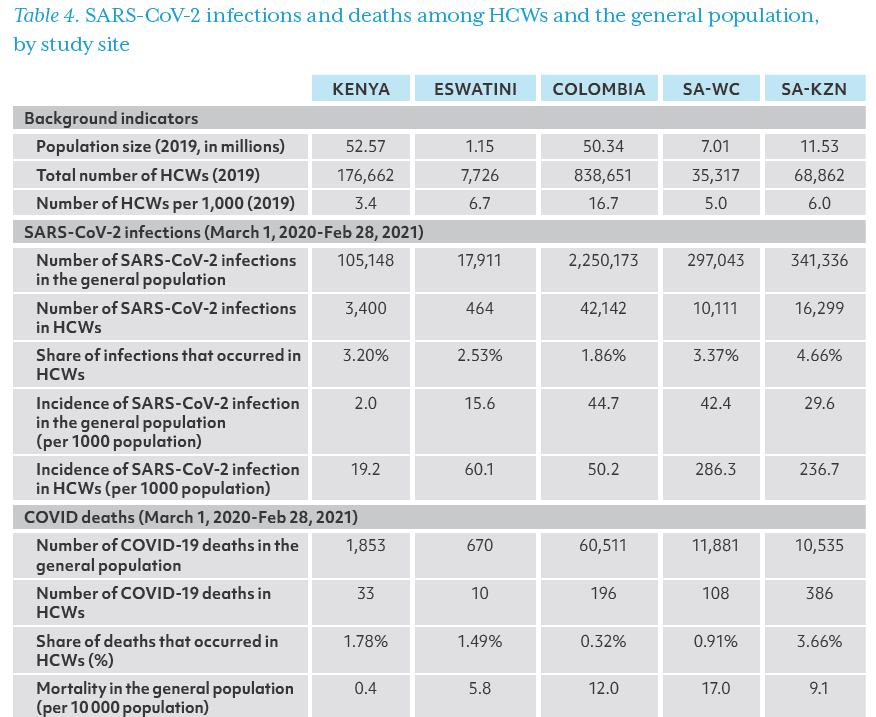
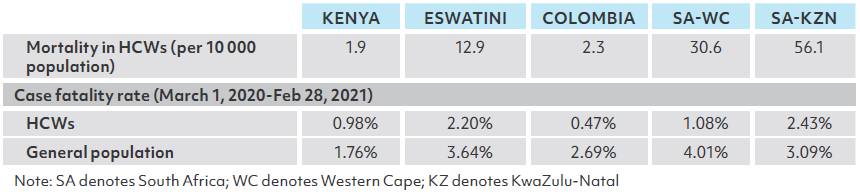
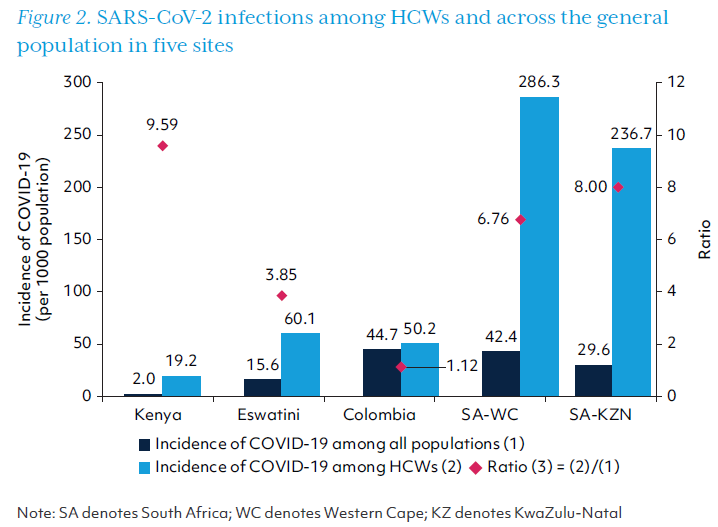
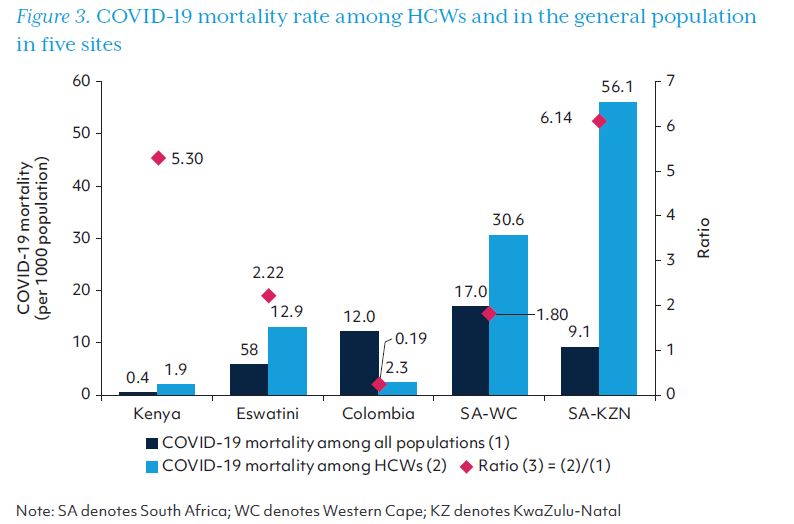
The number of SARS-CoV-2 infections and deaths among HCWs is compared with that among the general population in Table 4. KwaZulu-Natal province had the highest proportion of SARS-CoV-2 infections occurring in HCWs (4.66%), and Colombia had the lowest, 1.86%. The COVID-19 incidence among HCWs was higher than in the general population in all study sites – almost 10 times higher in Kenya and 7-8 times higher in the two provinces of South Africa. Unlike other study sites, the incidence among HCWs was only slightly higher than the general population (50.2 vs. 44.7) in Colombia which had the highest level of human resources for health measured by the HCW density. Mortality due to COVID-19 was higher for HCWs than the general population in each site except Colombia, which had the lowest case mortality rate as shown in Table 4. COVID-19 incidence and mortality rates in HCWs and the general population are also depicted in Figures 2 and 3.
I-B. Costs on Pathway 1
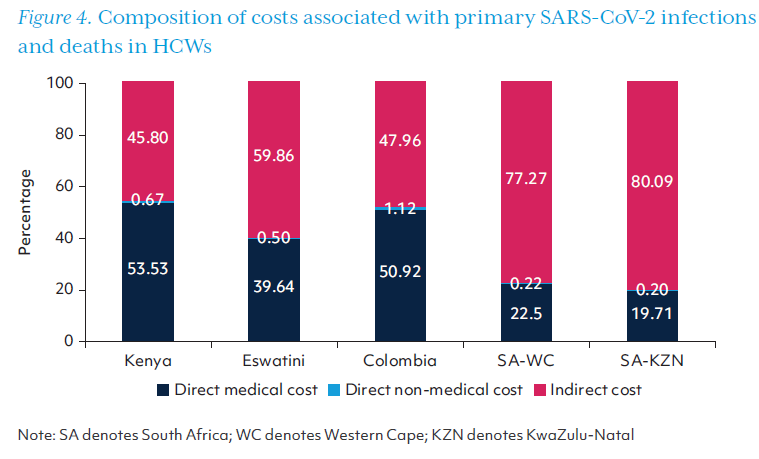
Table 5 presents the costs directly associated with HCWs who became unwell with COVID-19 disease, including their medical cost, non-medical costs related to seek ing care, along with the productivity losses due to morbidity and mortality (indirect costs). The costs accrued along Pathway 1 ranged from US$2.00 million in Eswatini to US$163.47 million in KwaZulu-Natal province alone, with a combined cost of US$252.29 million across both provinces in South Africa. Indirect costs comprised the largest share of costs associated with HCW infections and deaths in Eswatini, and KwaZulu-Natal and Western Cape provinces in South Africa (Figure 4).
II. Pathway 2: The economic costs of secondary infections and deaths due to transmission of SARS-CoV-2 from infected HCWs
II-A. Secondary infections and deaths due to transmission from infected HCWs
Table 6 shows the number of secondary infections and deaths due to HCW infections. There were an estimated 2,607 secondary infections in Eswatini and 9,939 in Kenya. Colombia, Western Cape, and KwaZulu-Natal had more than 40,000 secondary infections in each location, with 69,331 secondary cases in KwaZulu-Natal.
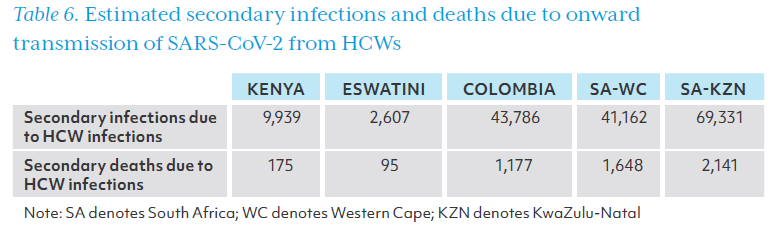
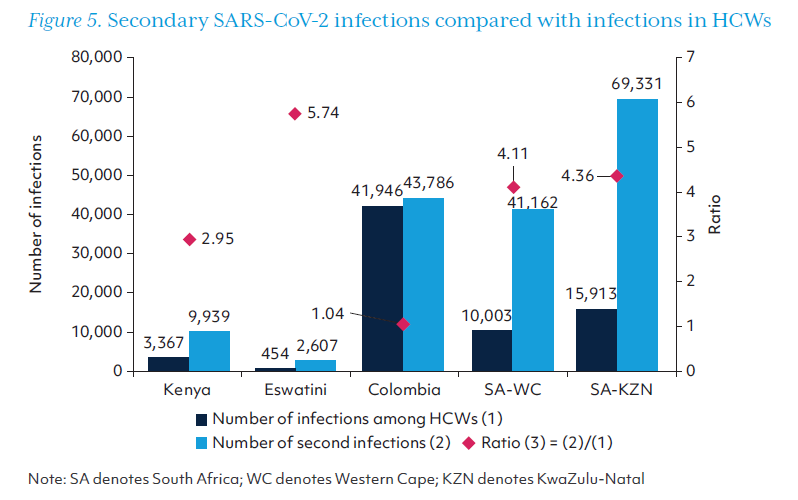
Compared with the number of SARS-CoV-2 infections in HCWs, there were at least three times as many secondary infections in all study sites except Colombia (Figure 5). Deaths due to secondary infections were greater than HCW deaths in every study site, with over 15 times as many deaths due to secondary infection as HCW deaths in Western Cape province. Secondary deaths are displayed in Figure 6 relative to deaths in HCWs.
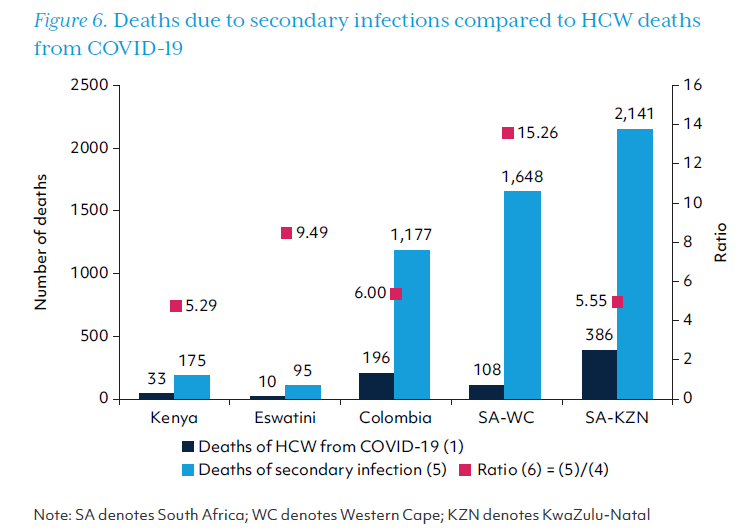
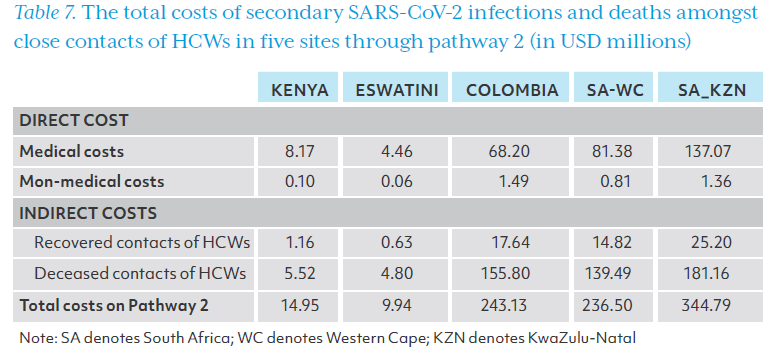
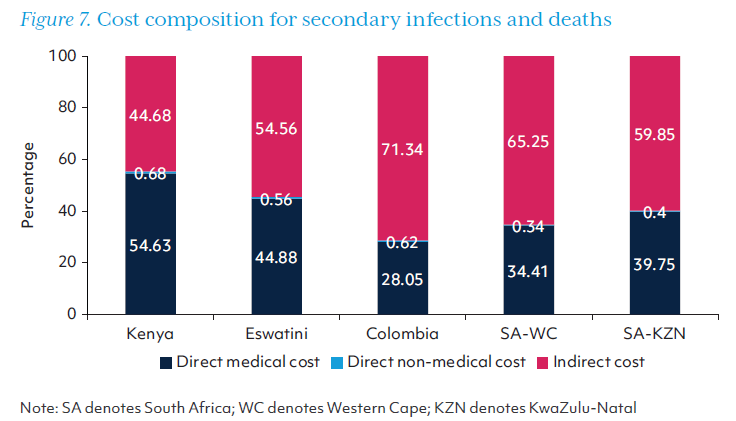
The direct and indirect costs of secondary SARS-CoV-2 infections and deaths are presented in Table 7. The Indirect cost due to the loss of productivity from both recovered and diseased COVID-19 patients transmitted by HCWs dominated the total cost through pathway 2. Along pathway 2, the cost of these infections and deaths in Eswatini was US$9.94 million, US$14.95 million in Kenya, and more than US$200 million for other study sites. As in pathway 1, the indirect costs accounted for about half of the total costs accrued via this pathway, and the share was even higher in Colombia, KwaZulu-Natal, and Western Cape (Figure 7).
III. Pathway 3: Economic burden associated with excess non-COVID mortality due to HCW infections
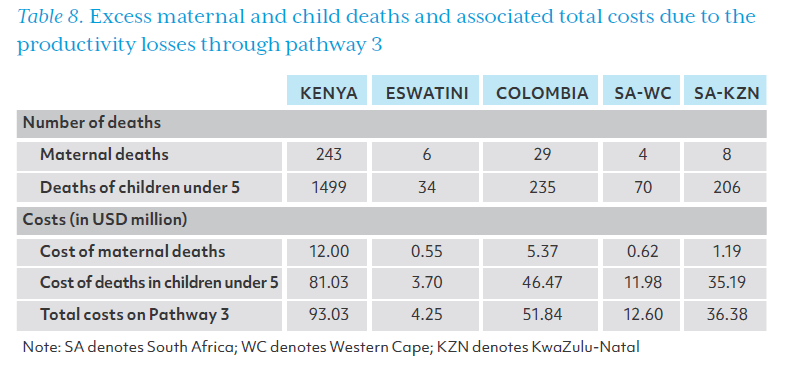
The costs incurred on Pathway 3 relate to excess maternal and child deaths resulting from HCW absenteeism that compromises health service delivery. The associated costs were included in Table 8. Excess maternal and child deaths in each country or province, along with the estimated total cost of productivity losses to society due to premature death, are included in Table 8. Maternal deaths were the least affected by HCW shortages in Eswatini and both provinces of South Africa. Kenya had the highest estimated excess maternal and child deaths, and the costs along this pathway were similarly highest in Kenya at US$93.03 million. In our analysis, Colombia had the second-highest increase in maternal and under-five mortality rates, amounting to US$51.84 million in economic losses along pathway 3.
IV. Total economic burden associated with SARS-CoV-2 infection in HCWs (all pathways)
The total economic burden of SARS-CoV-2 infection in HCWs along pathways 1, 2 and 3, as well as the estimated burden per HCW infection are displayed in Table 9. Economic losses ranged from $16.19 million in Eswatini to $544.64 million in KwaZulu-Natal province alone. The cost per HCW infection was more than $30,000 in four sites, while the cost per HCW infection was estimated to be $10,105 in Colombia. This equates to between 1.54 times (Colombia) to 17.98 times (Kenya) GDP/capita. The low cost per HCW infection in Colombia was primary due to the lower risk of HCWs transmitting SARS-CoV-2 to their close contacts.
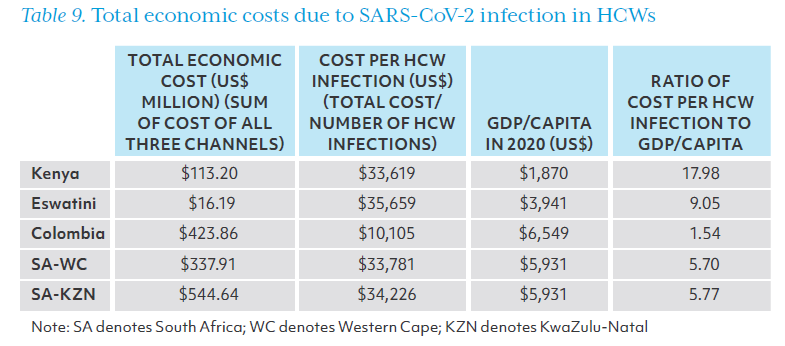
Figure 8 depicts these economic costs as a share of the annual total health expenditure (THE) and of annual public health expenditure (PHE) (both Government health expenditure and compulsory health financing schemes). These indicators showed the relative significance of the economic cost of HCWs infection, ranging from 1.51% in Colombia to more than 8% of annual THE in the two provinces of South Africa, and from 1.95% in Colombia to more than 17% of annual PHE in the two provinces of the PHE.
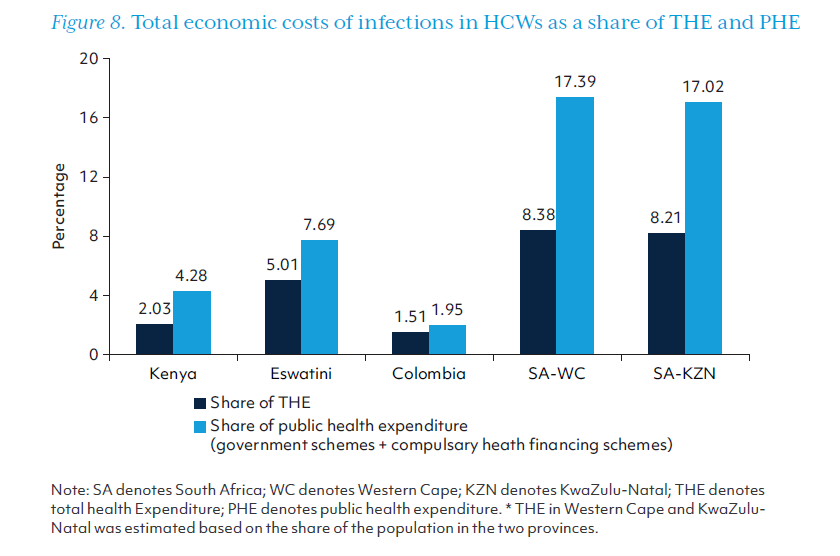
The five study sites show different patterns across the three pathways of economic costs. (Figure 9). In Kenya, the cost along pathway 3, which was associated with excess maternal and child deaths arising from HCW infections, accounted for the largest share of the total economic loss (82.2%), while the costs along pathway 2, which was associated with secondary SARS-CoV-2 infections and deaths, rep resented only 13.2% of the total cost. In Eswatini, Colombia, Western Cape, and KwaZulu-Natal, where more than 5% of HCWs had SARS-CoV-2 infection, the cost along pathway 2 (secondary infections and deaths) accounted for the majority (57.4%-70.0%) of the economic loss. In Colombia and both provinces of South Africa, the primary costs of HCW infections made up nearly a third of the total economic cost (pathway 1), while the cost associated with excess non-COVID mortality was relatively less substantial (pathway 3).
V. Scenario and sensitivity analysis
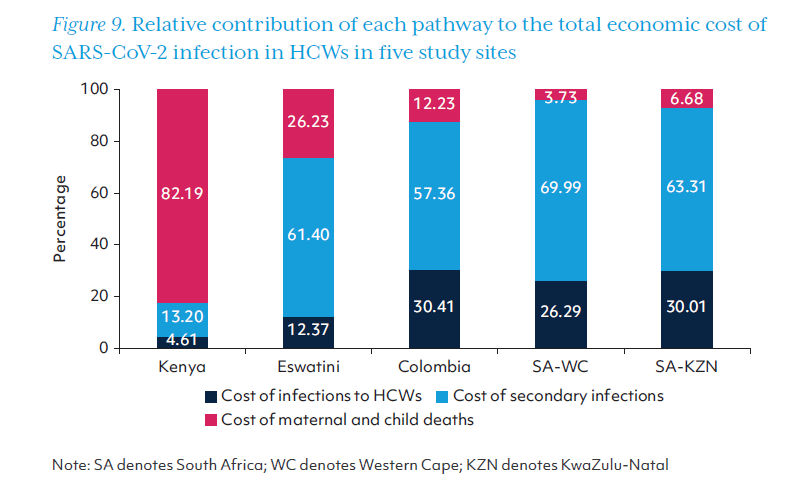
Table 10 shows the total economic cost and cost per HCW infection under the low impact and high impact scenarios. The total cost in Kenya ranged from $35.83 million to $246.12 million with the cost per HCW infection between $10,641 and $73,098. The two provinces in South Africa had a relatively smaller variation, and their cost per infection was between $28,032 and $39,793.
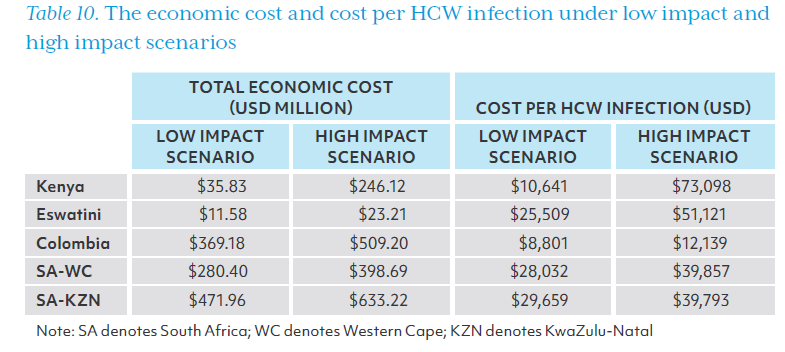
One-way sensitivity analysis shows the percentage change from the total economic costs we had calculated when four parameters were adjusted (Figure 10). Kenya was most sensitive to variations in the parameters used to cost pathway 3, while the South African provinces were most sensitive to variation in the proportion of inpatients who were close contacts of infected HCWs. The cost along pathway 2 could range from 14.1% lower to 13.3% higher than our estimate for Western Cape depending on this variable. The extent to which HCW productivity was impacted by HCW infections (pathway 3) affected the estimated results in Kenya and Eswatini substantially, particularly in Kenya. The estimated cost along pathway 3 could range in Kenya from 40.6% lower to 40.6% higher than the main analysis depending on this variable. The U5MR and MMR elasticities as they relate to HCW density had a larger impact in Kenya and Eswatini than the other three sites.

In this analysis, we have estimated the economic costs associated with SARS-CoV-2 in HCWs in three countries and two provinces of a fourth by calculating: (1) the primary costs of SARS-CoV-2 infections and deaths in HCWs themselves; (2) the costs of secondary infections and deaths due to transmission of COVID-19 from HCWs; and (3) the costs of excess maternal and child deaths arising from disrupted essential health services in the face of HCW shortages and diminished productivity.
SARS-CoV-2 infections in HCWs result in enormous societal costs, especially in country settings where HCWs face a disproportionate risk of infections compared with the general population. Our findings corroborate previous reports of increased risks of exposure and higher infection rates in HCWs compared to the general population from all income settings and regions. Countries in this study with substantial differences in the SARS-CoV-2 infection rate between HCWs and the general population (Kenya, Eswatini, and both provinces of South Africa) bore the greatest financial toll as a percentage of total public health expenditure (Figure 8), with a societal ‘price’ per HCW infection that is several times higher than the per capita GDP. Colombia had the lowest cost per HCW infection (Table 9). Infection rates among HCWs did not vary greatly from COVID-19 incidence in the general population, and baseline MMR and U5MR are low in Colombia (Annex 1). These findings highlight the critical importance of adequate infection prevention and control measures to minimize the risk of HCWs being infected and prevent such enormous cost incurring.
This study demonstrates the negative externality that HCW infections can cause; when HCWs are infected, the health and economic impacts of those infections go far beyond their individual health and livelihoods. Although the cost of HCW primary infections and deaths is not insignificant (pathway 1), the total cost is mostly related to onward infectious transmissions from sick HCWs and disruption of maternal and child health services as a result of HCW illness, isolation, or death. The substantial economic cost due to the service disruption is consistent with previous research estimating the enormous costs associated with HCW shortages in LMICs due to brain drain. These results demonstrate the need to closely monitor the situation of HCWs during epidemics and pandemics, to include and quantify the economic costs of HCW infections beyond costs incurred to HCWs themselves.
Maternal and child deaths related to compromised health service delivery greatly contribute to economic losses due to HCW infections, especially when countries have poor maternal and child health outcomes and inadequate human resources for health at baseline. Most maternal and child deaths are avoidable, and excess deaths due to disrupted maternal and child health services are tragic reversals in earlier progress. Excess maternal and child deaths are also economically costly: the costs of excess maternal and child deaths due to HCW infections amount to US$93 million and comprise the greatest proportion (82%) of total costs in Kenya. Of the countries and provinces included in this study, Eswatini and Kenya had the highest pre-pandemic U5MR and MMR, and our cost estimates in Kenya and Eswatini were sensitive to the U5MR and MMR elasticities relative to HCW density as well as changes in productivity for HCWs remaining in post.
In countries with high U5MR and MMR, there may be fewer other stopgaps in place to prevent unnecessary deaths when the health care workforce is compromised. Maternal and child mortality rates similarly rose in West African countries affected by the 2014 Ebola outbreak, which saw severely disrupted provision of essential services including child vaccination and antenatal care. Unfortunately, public health measures such as movement restrictions to ‘flatten the curve’ of COVID-19 outbreaks further disrupt essential health services including maternal and child health care. One modelling study estimated that COVID-19-related restrictions reduced delivery of essential maternal and child health services in LMICs by 9.8–51.9%, resulting in substantial excess maternal and child deaths globally. As maternal and child health services seem to be sensitive to workforce disruptions during public health emergencies, dedicated measures to safeguard maternal and child health in countries with high baseline mortality rates are critical. These might include task-shifting or bolstering child health with interventions that aren’t dependent on HCW density. The studied countries took different strategies to mitigate the service disruption. Colombia developed the pandemic containment plan, which included human resource retainment strategies. In Kenya, hiring new HCWs in the public sector was initiated although rehiring retirees was prohibited due to the concerns of their risk of infection. Nevertheless, the high estimated costs in Kenya due to excess maternal and child deaths as a result of disrupted health services again highlight the importance of protecting HCWs, minimizing disruptions to essential health service delivery, and maintaining service utilization levels, while urgent work to reduce maternal and child mortality continues globally.
These costs are likely to be conservative estimates of the true cost to society of SARS-CoV-2 infection in HCWs. Our economic cost estimates are likely to be conservative for several reasons. First, it is likely that the SARS-CoV-2 infections and deaths were underreported. Under-reporting is common for infectious diseases generally, but the challenges to accurately reporting SARS-CoV-2 infections and deaths are well recognized, including access to tests, testing capacity and the challenges in defining causality in the event of death. Second, SARS-CoV-2 infections and deaths among HCWs are likely to have longer-term impacts on the supply of the health workforce that we did not attempt to capture. For example, COVID-19 surges tend to prompt additional HCWs to leave their jobs in clinical care. Students or young people who have witnessed the disproportionate infectious risks faced by HCWs may change their career choices. Third, we focused on three pathways through which HCW infections incur economic costs. While these pathways likely comprise the most important sources of economic losses, we were not able to quantify (1) the costs associated with worse health outcomes beyond excess maternal and child mortality, especially in the area of non-communicable diseases which account for a significant share of disease burden in countries. Disruptions in essential health care services in the first year of the pandemic were pervasive, especially in LMICs, and certainly include impacts on services beyond maternal and child health care. (2) Nor does this analysis include the cost of training HCWs to replace those who are no longer working or alive as a result of SARS-CoV-2 infection. Training HCWs is costly and slow, and is one focus of ongoing efforts to ameliorate the global health workforce shortage. (3) This study does not account for the costs associated with mental health impacts of COVID-19 on HCWs or other long-term sequelae of infection such as long-covid. (4) The COI approach does not include the cost of interventions to prevent the illness, such as the deployment of COVID-19 vaccines and provision of PPE among HCWs.
Limitations. Several limitations of this study should be acknowledged. First, this study does not use an infectious disease transmission model to estimate the secondary infections from infected HCWs in each country. Instead, this study estimated the odds ratio of infection for close contacts of HCWs based on an epidemiological study in a high-income country and used a log-linear regression to adjust for the differences in the relative risks faced by HCWs in each country. The estimations thus may not fully reflect the relative risk of secondary transmission of SARS-CoV-2 through exposure to infected HCWs in different settings. Further epidemiological research is likely to enable the global public health community to estimate the risk of SARS-CoV-2 transmission more precisely in different settings.
Second, data on SARS-CoV-2 infections and deaths in HCWs for the two sites in South Africa does not include data from the private sector. Data from the other study sites represents composite information from both public and private sectors. If infections and deaths occurring in the private sector in Western Cape and KwaZulu were included in this analysis, the economic costs would be even higher for pathway 1. Limited evidence from Saudi Arabia suggests that the SARS-CoV-2 infection rate may be similar in public and private facilities. Assuming that this is also true for the five sites in this study, the economic costs along pathways 2 and 3 would not significantly vary.
Third, some data were not available in some study sites. In this event, we made assumptions or drew from the literature from neighboring countries. Taking the cost per meal and cost of travel in Eswatini as an example, we used the analogous prices for South Africa as proxies when calculating direct non-medical costs. These inaccuracies are thus not likely to substantively change our results, as we used the best available data to substitute for missing figures.
Fourth, in estimating the potential service disruptions due to the deaths and illness of HCWs, we neglect the potential strategies that countries had taken to mitigate the impact. Kenya hired more HCWs in the public sector and Colombia accelerated the validation of foreign qualifications and deployed medical students and graduates. Their impact on the estimated economic burden depends on the size of additional personnel adding to the health workforce.
Finally, we cannot precisely calculate the change in productivity of HCWs remaining in their post in the first year of the COVID-19 pandemic (pathway 3). Productivity could vary by the number of colleagues absent, the number of deaths in HCW colleagues or family members, mental health status, the presence of post-covid symptoms, and the change in patient demand. Instead, we made an arbitrary assumption that all these factors reduce the overall productivity by 10 percent. The sensitivity analysis shows that the results from Kenya are sensitive to this assumption. Even under the low impact scenario with a decrease in productivity of 5%, this still translated to US$10,641 per HCW infection, 5.7 times higher than per capita GDP.
Policy Implications
To accurately estimate the toll of the pandemic on HCWs in economic terms, countries and the global health community need to have high-quality data on HCW infections and deaths based on a consistent methodology. Infection rates among HCWs vary widely between countries, with estimates ranging from 1% to 30–35%, and up to nearly 50% in some locations. While methodologies for measuring deaths due to COVID-19 have been questioned, in general, SARS-CoV-2 infection and death rates in HCWs in the first year of the pandemic are widely understood to be under-reported and under-estimated. This has been attributed to incomplete data, inadequate access to or availability of testing, and the fact different municipalities took different approaches to case-finding, testing, and documenting COVID-19 cases and deaths amongst HCWs. In part due to limits on testing capacity, health systems in the first year of the pandemic prioritized testing of symptomatic HCWs and were delayed in implementing routine screening of asymptomatic HCWs. Multiple data sources in the decentralized health system and across public and private sector providers present an additional layer of complexity for compiling readily available national-level aggregate data.
Without accurate accounting of HCW infections and deaths, adequate steps cannot be taken to properly protect them. Conversely, quality data on HCW infections and deaths may help identify the most vulnerable professionals requiring additional protective measures.12 For decades, the International Labor Organization(ILO)’s Occupational Safety and Health Convention (1981) Article 11(c) has stipulated the need to establish and apply “procedures for the notification of occupational accidents and diseases, by employers.”78 More recently, the seventy-fourth World Health Assembly resolution A74/A/CONF./6 on Protecting, safeguarding and investing in the health and care workforce has further paved the way for action by specifying that national health workforce accounts should be utilized and expanded “for accelerating the continuous measurement and monitoring of the […] safeguarding and protection of the health and care workforce, including the collection of data pertaining to health and care workers’ morbidity and mortality, in the context of their work responding to epidemics and/or pandemics.”79 These provide a strong mandate for investing in infectious disease reporting methodologies that capture more accurate and complete data on HCW infections and deaths.
Transparency of accurately measured HCW infections and deaths due to COVID-19 can reduce risks to fellow HCWs and their contacts and minimize the social and economic toll of the pandemic. Failing to report HCW cases and deaths likely hindered awareness of the full gravity and impact of COVID-19 on the health care workforce and delayed actions for improvement, for both HCWs and patients, given that globally, health care-associated infections are the single largest risk to patient safety. Case numbers and HCW deaths were often not publicly available. When collecting data from other study sites, the infection and death data routinely collected by countries are not disaggregated by profession. In Kenya, data on HCW infections and deaths were made publicly available towards the end of 2020 and led to tensions between service providers and the authority. HCW infection rates may not have been made public out of concern that they would deter people from seeking essential health care services. Indeed, fear of becoming infected with SARS-CoV-2 due to exposure to a health care environment or infected HCWs is a well-described deterrent to health-seeking.
The economic costs associated with HCW infections are preventable if infectious risks to HCWs are mitigated upfront by working towards safe health facilities with full implementation of infection prevention and control (IPC) measures and water, sanitation and hygiene (WASH) standards. Epidemiologic reviews of outbreaks repeatedly cite the same exposure risks: lack of systematic IPC protocols and training on how to follow them, and lack of appropriate WASH standards and resources. The 2002–2003 SARS epidemic was made worse by continued IPC lapses after the disease was identified. A review of the MERS response attributed the improvement in infectious risks to enhanced IPC efforts, including regular training of healthcare workers in IPC and auditing of IPC in healthcare facilities. Studies on the correct use of adequate PPE have consistently demonstrated its effectiveness in reducing the spread of COVID-19 from HCWs to patients and to HCW colleagues. In light of these results, modeling that suggests high cost-effectiveness of providing PPE to HCWs is not surprising. Pre-service education and on-the-job training need to incorporate and regularly re-visit the training on IPC practices and correct use of PPE. For example, South Korea mandated regular PPE training for the health workforce after its 2015 MERS outbreak. There are robust IPC standards and normative occupational health guidance, reiterated by World Health Assembly resolution A74/A/CONF./6. Increased and coordinated investments in IPC training, supplies (including PPEs), and monitoring, including adequate WASH facilities, are needed to support the full implementation of IPC guidance; implementation research may also help in this regard.
Protecting HCWs during public health emergencies including pandemics is an integral part of a building back better agenda. The COVID-19 pandemic has again spurred countries and the global public health community to invest in and prioritize building resilient health systems. Key components of resilient health systems are adapting public health functions to mount a timely response to the infectious threat and protecting HCWs in order to preserve essential health service delivery. This study has described and quantified the economic impacts due to infections and deaths in the health workforce during the first year of the COVID-19 pandemic. It shows that the societal cost of SARS-CoV-2 infection in HCWs as a percentage of total health expenditure was highest in this analysis in the four study sites with the lowest HCW density. Protecting health workers needs to be embedded in all aspects of human resource production, deployment, and compensation. Well-developed hazard compensation policies, for example in Vietnam, demonstrate a recognition of the importance for overall health outcomes of protecting HCWs and maintaining an adequate healthcare workforce in times of crisis. Deployment of the health workforce during a crisis should be complemented by comprehensive support measures including physical protection, psychological support, and child/family support, many of which have been implemented during COVID-19 in many countries and require institutionalization for long-term impact. Given the importance of HCWs for enacting public health emergency responses – and the ways in which lower HCW density could render essential health services more vulnerable to disruption – policies to attract, retain, and motivate qualified HCWs should be placed at the center of building more resilient health systems in LMICs. Better protecting health care workers can also contribute to gender equality as women comprise 70% of the global health workforce and more than 80% of the nursing and midwifery workforce, the profession that is most vulnerable to infection risks.
The enormous negative externalities associated with HCW infections and deaths necessitate accountability for protecting HCWs during infectious outbreaks from governments, with guidance from the WHO and support from funders. Many countries adopted various strategies to protect HCWs during the pandemic. For example, the Kenya government reviewed and identified policy gaps (e.g. working environment monitoring) and implementation challenges of HCWs protection (e.g. shortage of PPE). Specific resources were allocated to address the gaps and challenges. In Colombia, actions, such as training on HCW protection, ensuring the availability of PPE, and vaccination, were taken to enhance the protection of HCWs from SARS-CoV-2 infection. The protection of HCWs need a holistic approach. In addition to setting policies to build and maintain a core health care workforce, government responsibilities for the health workforce encompass mobilizing and allocating resources required for protecting health workers, ensuring proper use of these resources, and being accountable to the results. We suggest actions to protect health care workers include, but are not limited to: improving collection of data pertaining to HCW infections and deaths; strengthening national IPC programs and supporting the full implementation of WASH standards; developing and deploying training, tools and resources for a safer health care workforce; reinforcing supply chains – including emergency procurement policies and local production – to maintain adequate supplies of PPE; equitably distributing countermeasures to HCWs, including quality-ensured PPE in sufficient quantity; reviewing laws and regulations to protect HCWs from attack and stigma; providing HCWs with mental health support and appropriate financial compensation; and improving overall transparency as it relates to the safety of the workforce and facilities. The Member State-led WHA resolution A74/A/CONF./6 on Protecting, safeguarding and investing in the health and care workforce enshrines the above recommendations, and together with ILO’s Occupational Safety and Health Convention (1981) Article 16, point to the need for a clear accountability framework to ensure HCWs are protected from potential health and financial risks in the setting of an infectious outbreak.
Conclusion
This study shows that SARS-CoV-2 infections and deaths in HCWs in the first year of the pandemic posed substantial economic burden on whole societies. Most of the economic costs go beyond infections and deaths in HCWs themselves. There is an opportunity now as countries begin to ‘build back better’ from COVID-19 to invest in and prioritize greater health system resilience, particularly in LMICs. Failing to enact protective measures oriented toward the health workforce would almost certainly incur additional, substantial costs in future waves of COVID-19 or future pandemics. Governments should take the lead in protecting HCWs.
ANNEX 1
Sources of key parameters for modeling, by country
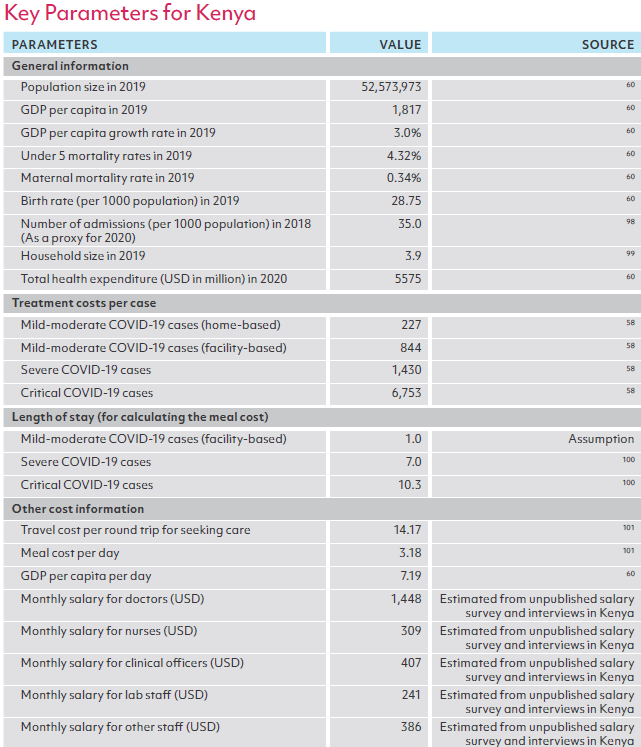
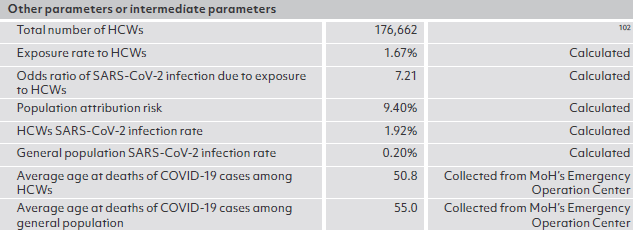
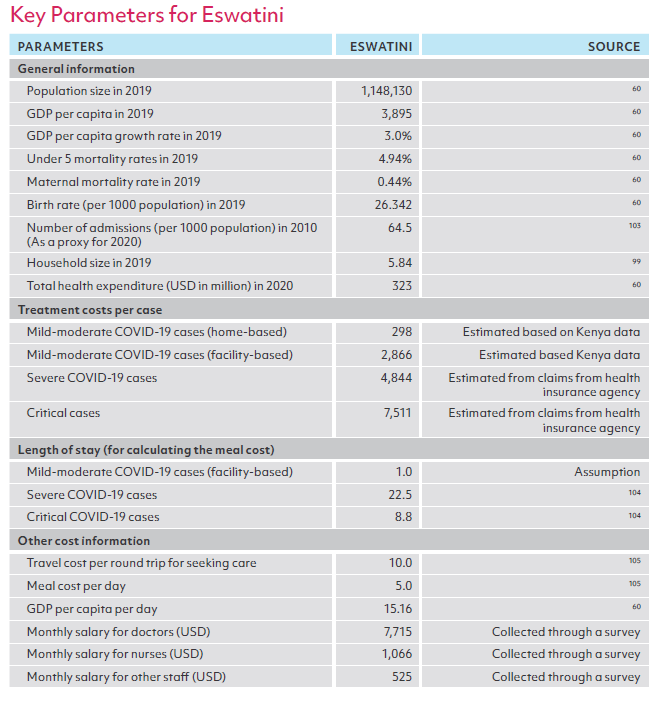
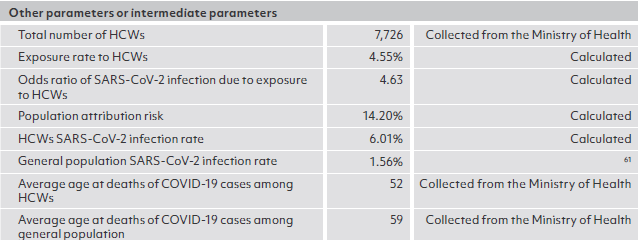
ANNEX 2
Estimation of exposure rate and odds ratio for secondary infection
To estimate Ei, we divided the sum of the average number of household contacts and patients admitted to health care facilities by the total population. To account for the difference in the duration and proximity of contact between HCWs and their household members compared to patients admitted to a health care facility, we assumed that inpatients had roughly 20% of the exposure to HCWs as household members.
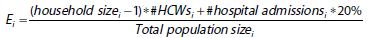
There is scarce literature on relative risk or odds ratio of SARS-CoV-2 infection due to HCW exposure. A national study in Scotland showing that family members of HCWs had a higher likelihood of SARS-CoV-2 infection, with an odds ratio of 1.79. By assuming that the higher odds ratio of infection amongst close contacts of HCWs has a log-linear relationship with the ratio of SARS-CoV-2 infections in HCWs to the general population in any given country, we estimated the odds ratio for other countries based on Scotland’s estimated odds ratio.

References







Acknowledgments